Adjunct Protocol for Effective Asthma Management
On this page
We introduce “KNOW YOURSELF”: adjunct protocol for effective asthma management.
Introduction
Asthma is a disease that affects lungs in children and adults. Asthma causes wheezing, breathlessness, chest tightness, and coughing at night or early in the morning.
The prevalence of asthma has nearly doubled over the last decades, especially in Westernized countries and developing countries with rapid urbanization. We don’t know all the things that can cause asthma, but we do know that genetic, environmental, and occupational factors have been linked to developing and managing asthma. There’s currently no cure for asthma, but mainstream and non-conventional treatments can help control the symptoms so you’re able to live a normal, active life.
Majority of asthma management plans focus only on optimization of asthma pharmacotherapy. However, 5–10 % of the asthmatic population respond poorly to high dose inhaled and then systemic glucocorticoids. These patients form a category of severe asthma associated with poor quality of life, increased morbidity and mortality, and constitutes a major societal and health care burden 1.
Factors of Effective Asthma Management
According to the guidelines stipulated by the Asthma Care guide from National Institutes of Health2, achieving and maintaining asthma control requires, in addition to pharmacotherapy, addressing external factors that may cause worsening symptoms, and helping patients learn self-management skills.
Being under the same treatment, you can affect asthma positively (or negatively) by diet. Food reactions are named as one of the comorbid conditions that contribute to asthma severity by the comprehensive Asthma Management Guide3. Therefore, diet is one of these external adjunct lifestyle factors that is well under the patient’s control but requires self-education.
If we understand these dietary factors, we can control and manage them, to the best of our ability! Thus, an important adjunct to an asthma management protocol is knowledge of the positive or negative effects of your dietary preferences.
Knowledge is power! Attain your power when you:
- Know YOUR foods. Find foods you are intolerant to, along with corresponding levels of intolerance, and avoid these foods for as long as it is needed.
- Know YOUR nutrients. Change diet and use supplements if there is a nutritional deficiency.
- Know YOUR genes. Use diet (and/or dietary supplements) to compensate genetic factors or mutations that make your asthma and/or COPD worse.
Let’s talk about foods first.

Role of the Diet
We consider diet as a sum of the foods consumed by the person. Our diet sustains our life. When our bodies do not work well with certain foods, a food intolerance (or food allergy) may happen. We introduced topic of Food Intolerance in this article.
It is estimated that 1/5 of the world population has adverse reactions to their foods. These reactions are on rise. Those foods our bodies do not agree with can cause serious damage!
In the study 4 of a clinic population of patients with severe asthma (Birmingham Heartlands Hospital, U. K.), around two thirds reported at least one food which could exacerbate their asthma. 60% of the total clinic population have subsequently undergone food challenge studies and around 50% patients showed positive responses to one or more foods, either immediate or delayed.
Food Intolerance in Lung Disease Management
All adverse reactions to foods can be classified according to their underlying pathophysiology as the following 5:
- Toxic (ingested food toxins, rather infrequent): i.e. scombroid syndrome from the ingestion of spoiled fish containing a lot of histidine which is transformed into histamine in the gut by bacteria.
- Non-toxic
Immune-mediated (food allergy) reaction to foreign protein accompanied by release of allergy mediators
- IgE-mediated immediate reactions
- non-IgE (i.e. IgG, IgA, IgM, cell, or immune complexes) mediated delayed reactions
To prevent food allergy manifestations in asthma, you need to reduce the amount of histamine circulating in your body. The article “Histamine Intolerance: Am I Crazy or Allergic?” explains how excessive histamine (aka “histamine intolerance”) could bring havoc to your body.
You may want to start by reducing of amount of histamine your body releases. Substances calming down your cells histamine release are called mast cell stabilizers. Quercetin is a substance whose stabilizing function is at par or even better than prescription medications 6. Seeking Health provides trusted, made-in-the-USA, and properly tested supplements, including Gut Nutrients blend with Quercetin.
You continue by improving your ability to metabolize released histamine. DAO (Diamine Oxidase) in your gut is responsible for histamine metabolizing. Seeking Health offers THE BEST on the market DAO supplement, for you to able to eat and drink again! To get these supplements, just click on the images below and use code IQ10 for 10% discount at checkout.
Non-immune mediated (food intolerance)
- Enzymatic, resulting from enzymatic defects of the gastrointestinal tract i.e. lactose or gluten intolerance
- Pharmacological, caused by vasoactive amines (dopamine, histamine, norepinephrine, phenylethylamine, serotonin and tyramine) and other substances present in foods, all of which manifest pharmacological activity. In most cases, there is a dose-effect relationship. I.e. foods which contain high quantities of histamine may provoke symptoms similar to those of an allergic reaction, but normally, histamine is rapidly inactivated by enzyme di-aminoxidase (DAO) in the gastroenteric tract.
- Undefined, resulting from non-identified mechanisms, such as food additive reactions. Those who is predisposed may show intolerance reactions to sulphites, nitrites, nitrates, monosodium glutamate and some colourings. Possible symptoms are asthma, rhinitis, urticaria, itchiness, and migraines.

Managing adverse food reactions is a significant part of the lung disease control. It consists of managing the immune and non-immune factors contributing to asthma exacerbations.
Food allergy plays huge role in asthma. Approximately 50% of asthmatic children that required intubation for severe asthma had food allergy compared with about 10% of asthmatics not requiring intubation 4. Immune-mediated reactions are usually identified by an allergist using corresponding skin and serological tests. To maintain quality of life and avoid asthma exacerbations, allergy medications along with food avoidance are recommended 7.
Non-immune mediated food intolerance reactions (sometimes called metabolic) are more volatile and even borderline psychosomatic (see our article What is Food Intolerance). In general, measures to avoid food culprits result in considerable improvement in asthma symptoms, and reduction in drug therapy and hospital admissions 5. The problem is how to reliably identify these offensive foods, with high degree repeatability, and figure out minimal avoidance period to prevent intolerances (and thus asthma and COPD) from reoccurrence. This is a significant area of the lung disease management which more often than not is going unattended.
Let us sum that up. If you have asthma (or COPD), most likely there are some foods you have an adverse reaction to. While working with allergist can help to find allergy-causing foods and get medications to control the ill effects, there may be residual adverse food reactions that impede the lung disease management. Since pharmacological drugs did not improve them, most likely these are food intolerance reactions. But you are on your own when managing the food intolerances. The trickiest part is that food intolerances can come and go depending on your diet and lifestyle.
Controlling this aspect of your diet and lifestyle this is a lengthy process that requires strict discipline. Exclusion protocols literally can take years and bring minimal (if not none) outcomes.
To control disease, YOU need to take the reins! We administer and follow-up the North American-tailored version of Daphne LabⓇ BioMetaTests (Italy) that exclusively available through Montreal-based company Fresh Start Testing™. One test (approximately 2 weeks turn-round) assesses your body’s sensitivity to 1,100+ foods, including about 300 North American/Canadian food items!

Daphne LabⓇ BioMetaTests:
- satisfy needs of 90,000+ clients annually, in more than 80 countries
- are used in Italian hospitals
- are ISO-compliant
- utilize patented bio-spectrophotometry methodology
- have 94% repeatability and reliability rate
- are administered only through trained professionals
Gut Microbiome
It has been estimated that the human gut is populated with up to 100 trillion microbes. There has been an exponential increase in the evidence linking the gut microbiota’s dysbiosis with factors affecting asthma onset and control 8.
Butyrate and propionate are microbial metabolites of gut microbiota, specifically, by Ruminococcus, Clostridium, Eubacterium, Coprococcus, Veillonella, Bacteroides, and Acidaminococcus. These microbial metabolites have inhibitory effects on histone deacetylases that may promote the development of peripherally induced regulatory T-cells 9 triggering airway inflammation.

Another species worth mentioning is Lactobacillus rhamnosus that prevents airway function deterioration and promotes gut microbiome resilience10. Lactobacillus reuteri has a role in T-reg cells regulation alleviating airway inflammation11.
These and other beneficial probiotics are affected by the diet. To find out gut microbiome major players, along with diet augmentation ideas and levels of important enzymes affecting food intolerances, we also can utilize Daphne LabⓇ BioMetaTests.
Nutrient Deficiency and Excess in Asthma Management
Most often, your doctor would advise to top up your vitamin D intake. There were multiple trials to confirm vitamin D importance in asthma management, for example 12. Among adults with asthma and vitamin D deficiency, supplementation with weekly oral calcifediol compared with placebo improved asthma control over 6 months.
Vitamin D action is due to its effects on the immune system and genetic regulation of asthma susceptibility genes. According to this recent review 13 and sensitivity analysis, there is a statistically significant association between FokI gene polymorphism and TaqI gene polymorphism with asthma risk. In other words, being deficient on vitamin D genetically diminishes your chances to succeed in asthma management.
Review 6 analyzed correlation of the levels of several groups of nutrients with asthma management success.
Sodium excess
With exercise-induced asthma, reducing sodium intake may be beneficial in improving lung function.
Deficiency of Antioxidants (vitamins A, E, C, and minerals Zinc and Selenium)
Dysregulation of the oxidant-antioxidant balance can contribute to lung disease development and progression. Oxidative stress is one of the hypothesised mechanisms implicated in asthma pathogenesis. Individuals with asthma have been shown to have increased generation of reactive oxygen species compared to healthy controls, suggesting that modifying intakes of antioxidant compounds (such as vitamins, A, C, Zinc, Selenium etc) may be useful.
To restore antioxidant balance, NAC (n-acetyl-cysteine) is indispensable. It is a stable, sulphurcontaining form of the non-essential amino acid l-cysteine playing a fundamental role in the defense of the organism against damage caused by oxidative stress.
Omega-3 fatty acids deficiency
Several of inflammatory pathways are involved in asthma and airway hyperresponsiveness:
- inflammatory cytokine production
- production of inflammatory mediators regulated by omega-6 fatty acids
Omega-3 polyunsaturated fatty acids (EFA, or Essential Fatty Acids) include eicosapentaenoic acid, docosapentaenoic acid and docosahexaenoic acid. They are commonly found in significant amounts in marine sources such as salmon, herring and sardines. They are also well known for their anti-inflammatory properties that include inhibiting inflammatory cytokine production and competing with pro-inflammatory omega-6 fatty acids. Hence, supplementation with omega-3, if deficient, has been proposed as an adjunct in the treatment protocols of asthma.
To maintain Omega-3 status, get fish oil supplement. We recommend supplements from Seeking Health that are trusted, made-in-the-USA, and properly tested supplements. To get these supplements, just click on the images below and use code IQ10 for 10% discount at checkout.
Immediate assessment of Nutrient Status
This assessment brings cost-effective instant answers when you:
- Have poor results with “gold standard” asthma management protocols
- Feel stressed, tired, depressed
- Want to achieve optimal results

Nutri-IQ™ helps easy and conveniently identify nutritional gaps or toxicity as possible causes for your health problems, and especially lung disease. We will be happy to administer virtually the Nutri-IQ tests, and discuss with you test findings and their possible effect on your lung disease management.
Hormonal Insufficiency

In addition to these well-known respiratory symptoms, asthmatic patients also report that they experience tiredness, lack of energy, and daytime sleepiness 14. These problems negatively affect the quality of life and are accepted as relevant clinical symptoms.
This is mostly a secondary adrenal insufficiency in the patients treated with corticosteroid medications, “the golden standard” of asthma management protocols 15.
Alternative protocols often recommend adrenal gland extracts, to make life more plausible. We highly appreciate the help with normal stress hormone levels and energy provided by Seeking Health in this regard (do not forget to use 10% discount code IQ10 at checkout!)
Effect of Epigenetics on Asthma Management
We cannot change the genes we were created with, but we can minimize devastating effects of genetic problems through lifestyle changing. Actually, dietary way of asthma management is a very effective way to affect asthma triggers and overall control – and it affects negative effects of your genetic makeup too.
While genetic changes can alter which protein is made, epigenetic changes affect gene expression to turn genes “on” and “off.” The term epigenetics, derived from the Greek epi and genome, was introduced by Waddington to describe changes in phenotype without changes in genotype (i.e. changes in function without changes in structure). According to Wikipedia 16, Epigenetics are heritable characteristics that affect gene expression without altering DNA sequence, in contrast to genetics.
Epigenetic modifications occur during prenatal development, early childhood and adolescence. These are the life periods when people are susceptible to several asthma triggers. Prenatal environmental factors such as family smoking, and factors after birth such as traffic-related or air pollution, nutrients and drugs could be the triggering factors of epigenetic changes.
Several epigenetic mechanisms have regulatory role in immune responses and gene expressions that lead to asthma onset. Up-to-date, the following were identified:
DNA methylation
Methylation is regulated by the MTHFR gene mutation which found in asthmatic subjects 17. Epigenetic studies of childhood asthma 18 have identified specific methylation signatures associated with allergic inflammation in the airway and immune cells, demonstrating a regulatory role for methylation in asthma pathogenesis.

- For instance, genes implicated in airway remodeling (COL15A1, RB1, FOXP1, CCDC19), surfactant secretion (ACOT7, PPT2) and nitric oxide production in airways (ACP5), as well as genes associated with cytokine production and signaling (IL5RA, DICER1) and phagocytosis in blood (SERPINC1) are characterized by decreased methylation in asthmatic subjects.
- A subset of lymphocytes B produces IgE in allergic asthma and the most important genes associated with antigen presentation and IL-4 signaling such as CCDC80, DAPK3, LOXL1, PROC, FUCA2, SP100, ITCH, present increased methylation.
- A separate study identified increased methylation in the FOXP3 (genes that are involved in regulating the immune system) locus of asthmatic children exposed to higher ambient pollution and an association with impaired regulatory T cells
Thus, knowing MTHFR gene polymorphism may render help in asthmatic subjects.
Environmental influences play a significant role in asthma and have an effect on DNA methylation. Several studies like epigenome-wide association studies, and experimental and observational projects, have been conducted and published the previous years in an effort to clarify the interaction between environment and specific immune pathways that undergo epigenetic regulation.
Common symptoms of too much methylation include:
- headaches
- irritability
- insomnia
- runny nose
- body pain
- itchy skin
If you want to gently optimize your methylation (e.g. if you are too sensitive to most methylation support supplements), consider B Complex MF from Seeking Health.

Other important genes
Another interesting gene is COMT. COMT is responsible for the metabolism of catechol drugs 19 used in the treatment of asthma (Epinephrin, Racepinephrine, Fenoterol, Reproterol, Orciprenaline/Metaproterenol) 20.
Polymorphisms in DAO gene and corresponding enzyme deficiency are responsible for exacerbations of asthma/exercise induced asthma 21.
It is highly likely that environmental exposures interact with GST 22, or Glutathione S-Transferase gene involved in the antioxidant pathway. According to current knowledge, carriers of GSTM1(rs366631)/T1(rs17856199) null genotypes and GSTP1 Val105 (rs1695) genotypes are more susceptible to environmental oxidative exposures and have a higher risk of asthma.
NOS3 gene 23 was found to be almost un-methylated when children with asthma were subjected to high pollution conditions, strongly suggesting an epigenetic mechanism.
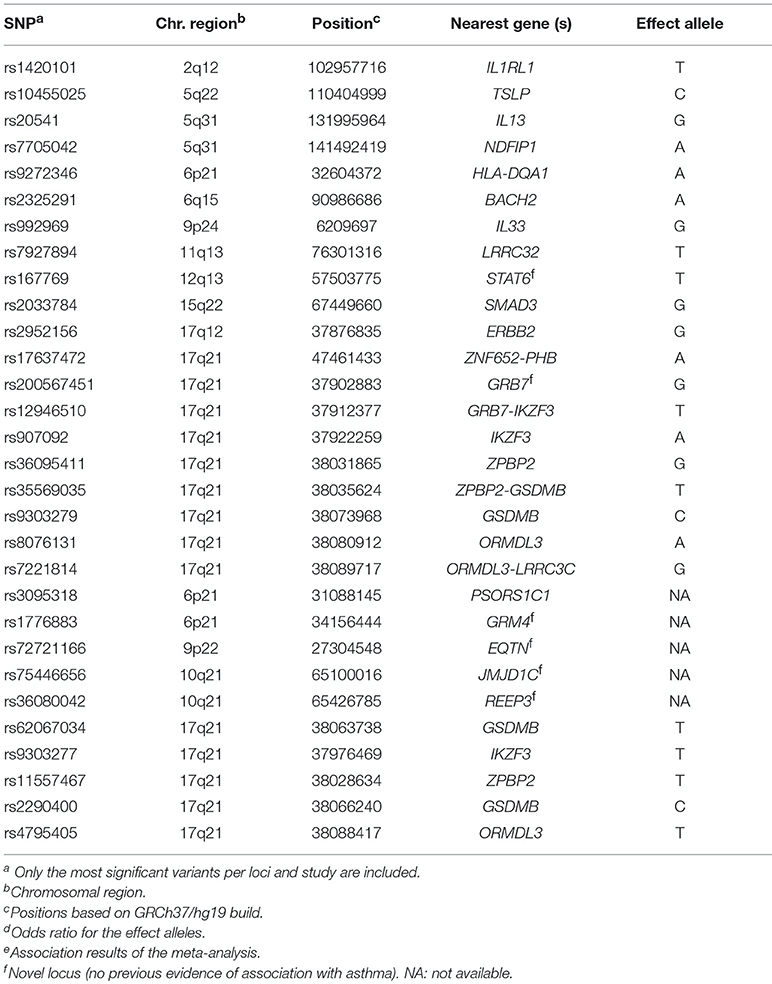
Up-to-date analysis of the most significant variants identified by the genome-wide association studies of asthma susceptibility can be found at 2019 publication 24.
Post-translational histone modifications
Histones are basic proteins abundant in lysine and arginine residues in eukaryotic cell nuclei 25 DNA is packaged by core histones to form an organized chromatin structure. Why histone modifications are important? 26
- histone methylation is associated with T-cell differentiation
- histone deacetylation by histone deacetylase (HDAC) results in gene silencing. HDACs are implicated in T-cells development and their inhibition could lead to allergic airway diseases. Moreover, defective HDAC2 activity is found in corticosteroid-insensitive severe asthma phenotypes.
- histone acetylation by histone acetyltransferase (HAT) usually results in a loose structure of chromatin, easily accessible to transcription factors that leads to the activation of gene expression.
MicroRNA (MiRNA) expression
miRNAs are approximately 20 nucleotides long non-coding. According to 27 microRNA are small, single-stranded, non-coding RNA molecules containing 21 to 23 nucleotides. They can be found in plants, animals and some viruses, miRNAs are involved in RNA silencing and post-transcriptional regulation of gene expression. Among miRNAs we can see 21 that:
- miRNA-19 is upregulated in the epithelium of severe asthmatic patients, targets TGFB2 mRNA, thus contributing to airway remodeling
- in peripheral blood of asthmatics, miR-625-5p, miR-22-3p, and miR-513a-5p were downregulated compared to controls.
Factors triggering epigenetic changes
The prevalence of asthma has nearly doubled over the last decades, especially in Westernized countries and developing countries with rapid urbanization. Your environment and modifiable risk factors, such as diet and exercise, can result in epigenetic changes, in contrast to genetics 28.
Nutrigenomic research, which examines the effects of food and its constituents on gene expression, shows that — like a number of pharmaceutical products — food can have beneficial effects or have adverse (side) effects depending on a person’s genotype 29. It is mind-blowing to understand, first time in your life, how your diet can affect gene expression and especially methylation!
On the market, there is an enormous number of companies doing different types of genetic testing, and we had written about them in our article Genetic Testing for Nutritional Intervention. We however are recommending one only. Epigenetic tests from Canadian research company Nutrigenomix examine your genes to determine if they can trigger food intolerances and other problems, like faulty methylation. These epigenetic tests are the BEST on the market!
- analysis of 77 genes and more than double polymorphisms (most companies do about 30 genes and about 100 polymorphisms)
- insights into methylations and asthma specifics (MTHFR, COMT, GST, NOS3 genes)
- insights into nutrient metabolism, food intolerances and sensitivities
- confirmed 99% accuracy of the tests (most companies do not confirm accuracy)
- analyzed in Toronto lab using state-of-the-art genetic testing procedures

We will be glad to help you to get Nutrigenomix test done, explain findings, implement recommendations, and even assist with creating the daily menus.
Please note that these tests also only can be ordered through trained professionals.
Conclusion: Adjust Your Diet!
We call the knowledge you can attain “KNOW YOURSELF “ protocol. And like any knowledge, it is useful only if applied – in our case, via diet modifications.
Creation of a clean diet and shopping for it may be a daunting task. Luckily, when you know what you can safely eat and how your current diet needs to be tweaked, you can meet your dietary goals without further complications. Act like a professional, and use professional services!
One of these services is That Clean Life. Build your customized meal plan for just $1/day (and you also can help others to eat plant-based, low histamine and salicylate, cancer support, antiinflammatory, … you name it!) This less-than-a-cup-of-coffee investment can make a huge difference in your life!

Another suggestion is eating locally grown clean food that did not spend forever in a warehouse or en route. Find services that get organic meats and poultry right to your door. One of the services we can recommend is truLocal, a North American local meat supplier. Use code TRUNUTRIIQ to get 3 lb of clean ground beef – FREE!

Let’s conquer asthma!
References
- Henderson I, Caiazzo E, McSharry C, Guzik TJ, Maffia P. Why do some asthma patients respond poorly to glucocorticoid therapy? Pharmacol Res. 2020 Oct;160:105189. doi: 10.1016/j.phrs.2020.105189.
- National Institutes of Health. Guidelines from the National Asthma Education and Prevention Program. https://www.nhlbi.nih.gov/sites/default/files/media/docs/12-5075.pdf Accessed on December 8, 2022
- UpToDate: An overview of Asthma Management. https://cmapspublic3.ihmc.us/rid=1VFF6SDN4-284NKHJ-13SV/An%20overview%20of%20asthma%20management.pdf Accessed on December 7, 2022
- C BAKER, J.G AYRES, Diet and asthma, Respiratory Medicine, Volume 94, Issue 10, 2000, Pages 925-934. https://doi.org/10.1053/rmed.2000.0873.
- Claudio Ortolani, Elide A. Pastorello, Food allergies and food intolerances, Best Practice & Research Clinical Gastroenterology, Volume 20, Issue 3, 2006, Pages 467-483, https://doi.org/10.1016/j.bpg.2005.11.010.
- Mlcek, J.; Jurikova, T.; Skrovankova, S.; Sochor, J. Quercetin and Its Anti-Allergic Immune Response. Molecules 2016, 21, 623. https://doi.org/10.3390/molecules21050623
- Isobel Stoodley, Lily Williams, Cherry Thompson, Hayley Scott, Lisa Wood Evidence for lifestyle interventions in asthma Breathe 2019 15: e50-e61; DOI: 10.1183/20734735.0019-2019
- Marchesi, J.R.; Adams, D.H.; Fava, F.; Hermes, G.D.A.; Hirsch, G.M.; Hold, G.; Quraishi, M.N.; Kinross, J.; Smidt, H.; Tuohy, K.M.; et al. The gut microbiota and host health: A new clinical frontier. Gut 2016, 65, 330–339.
- Benjamin T. Prince, Mark J. Mandel, Kari Nadeau, Anne Marie Singh, Gut Microbiome and the Development of Food Allergy and Allergic Disease, Pediatric Clinics of North America,Volume 62, Issue 6, 2015, Pages 1479-1492, https://doi.org/10.1016/j.pcl.2015.07.007.
- Irina Spacova, Wannes Van Beeck, Sven Seys, Fien Devos, Jeroen Vanoirbeek, Jozef Vanderleyden, Jan Ceuppens, Mariya Petrova & Sarah Lebeer (2020) Lactobacillus rhamnosus probiotic prevents airway function deterioration and promotes gut microbiome resilience in a murine asthma model, Gut Microbes, 11:6, 1729-1744, DOI: 10.1080/19490976.2020.1766345.
- Lingzhi Li, Zhifeng Fang, Zongmin Liu, Jianxin Zhao, Hao Zhang, Shunyu Wang, Jianxin He, Wenwei Lu, Wei Chen, Lactobacillus reuteri CCFM1072 and CCFM1040 with the role of Treg cells regulation alleviate airway inflammation through modulating gut microbiota in allergic asthma mice, Journal of Functional Foods, 76, 2021, 104286, https://doi.org/10.1016/j.jff.2020.104286.
- Andújar-Espinosa R, Salinero-González L, Illán-Gómez F, et al Effect of vitamin D supplementation on asthma control in patients with vitamin D deficiency: the ACVID randomised clinical trial Thorax 2021;76:126-133.
- Masoud Hassanzadeh Makoui, Danyal Imani, Morteza Motallebnezhad, Maryam Azimi, Bahman Razi, Vitamin D receptor gene polymorphism and susceptibility to asthma: Meta-analysis based on 17 case-control studies, Annals of Allergy, Asthma & Immunology, Volume 124, Issue 1, 2020, Pages 57-69, https://doi.org/10.1016/j.anai.2019.10.014.
- Van Herck, M.; Spruit, M.A.; Burtin, C.; Djamin, R.; Antons, J.; Goërtz, Y.M.J.; Ebadi, Z.; Janssen, D.J.A.; Vercoulen, J.H.; Peters, J.B.; et al. Fatigue is Highly Prevalent in Patients with Asthma and Contributes to the Burden of Disease. J. Clin. Med. 2018, 7, 471. https://doi.org/10.3390/jcm7120471
- Kachroo, P., Stewart, I.D., Kelly, R.S. et al. Metabolomic profiling reveals extensive adrenal suppression due to inhaled corticosteroid therapy in asthma.Nat Med 28, 814–822 (2022). https://doi.org/10.1038/s41591-022-01714-5
- Wikipedia. Epigenetics. https://en.wikipedia.org/wiki/Epigenetics Accessed on Dec. 5, 2022
- Li M, Tang Y, Zhao EY, Chen CH, Dong LL. Relationship between MTHFR gene polymorphism and susceptibility to bronchial asthma and glucocorticoid efficacy in children. Zhongguo Dang dai er ke za zhi = Chinese Journal of Contemporary Pediatrics. 2021 Aug;23(8):802-808. DOI: 10.7499/j.issn.1008-8830.2105035. PMID: 34511169; PMCID: PMC8428913.
- Gomez, J.L. Epigenetics in Asthma. Curr Allergy Asthma Rep 19, 56 (2019). https://doi.org/10.1007/s11882-019-0886-y
- Jiaojiao Li, Chunxiao Sun, Wenwen Cai, Jing Li, Barry P. Rosen, Jian Chen, Insights into S-adenosyl-l-methionine (SAM)-dependent methyltransferase related diseases and genetic polymorphisms, Mutation Research/Reviews in Mutation Research, Volume 788, 2021, 108396, https://doi.org/10.1016/j.mrrev.2021.108396.
- Drugbank. https://go.drugbank.com/categories/DBCAT000419 Accessed on December 7, 2022.
- Refaat, MM, Abdel-Rehim, AS, Elmahdi, AR, Mohamed, NA, Ghonaim, SS. Diamine oxidase enzyme: a novel biomarker in respiratory allergy. Int Forum Allergy Rhinol. 2019; 9: 1478– 1484.
- Dai, X., Bui, D.S. & Lodge, C. Glutathione S-Transferase Gene Associations and Gene-Environment Interactions for Asthma. Curr Allergy Asthma Rep 21, 31 (2021). https://doi.org/10.1007/s11882-021-01005-y
- Ji, N., Fang, M., Baptista, A. et al. Exposure to traffic-related air pollution and changes in exhaled nitric oxide and DNA methylation in arginase and nitric oxide synthase in children with asthma. Environ Health 20, 12 (2021). https://doi.org/10.1186/s12940-020-00678-8
- Hernandez-Pacheco Natalia, Pino-Yanes Maria, Flores Carlos, Genomic Predictors of Asthma Phenotypes and Treatment Response. Frontiers in Pediatrics, 7, 2019 10.3389/fped.2019.00006
- Wikipedia. Histone https://en.wikipedia.org/wiki/Histone Accessed on December 5, 2022
- Ntontsi P, Photiades A, Zervas E, Xanthou G, Samitas K. Genetics and Epigenetics in Asthma. International Journal of Molecular Sciences. 2021; 22(5):2412. https://doi.org/10.3390/ijms22052412
- Wikipedia. MicroRNA. https://en.wikipedia.org/wiki/MicroRNA Accessed on Dec. 4, 2022
- Bédard A, Li Z, Ait-hadad W, Camargo CA Jr., Leynaert B, Pison C, Dumas O, Varraso R. The Role of Nutritional Factors in Asthma: Challenges and Opportunities for Epidemiological Research. International Journal of Environmental Research and Public Health. 2021; 18(6):3013. https://doi.org/10.3390/ijerph18063013
- Laing, B.B.; Lim, A.G.; Ferguson, L.R. A Personalised Dietary Approach—A Way Forward to Manage Nutrient Deficiency, Effects of the Western Diet, and Food Intolerances in Inflammatory Bowel Disease. Nutrients 2019, 11, 1532. https://doi.org/10.3390/nu11071532